
On July 6, 2020, in response to numerous protests and protests by community activists, Johnson&Johnson Pharmaceutical Corporation announced a price reduction of $1.50 per day for bedaquiline, an anti-TB drug.
The news of this uniquely strengthens the hope of millions of people around the world who have MDR-TB and opens up a more visible opportunity to receive life-saving treatment for more patients. Note that the last petition urging the corporation to lower the price was signed by more than 120.7 thousand people from all over the world.
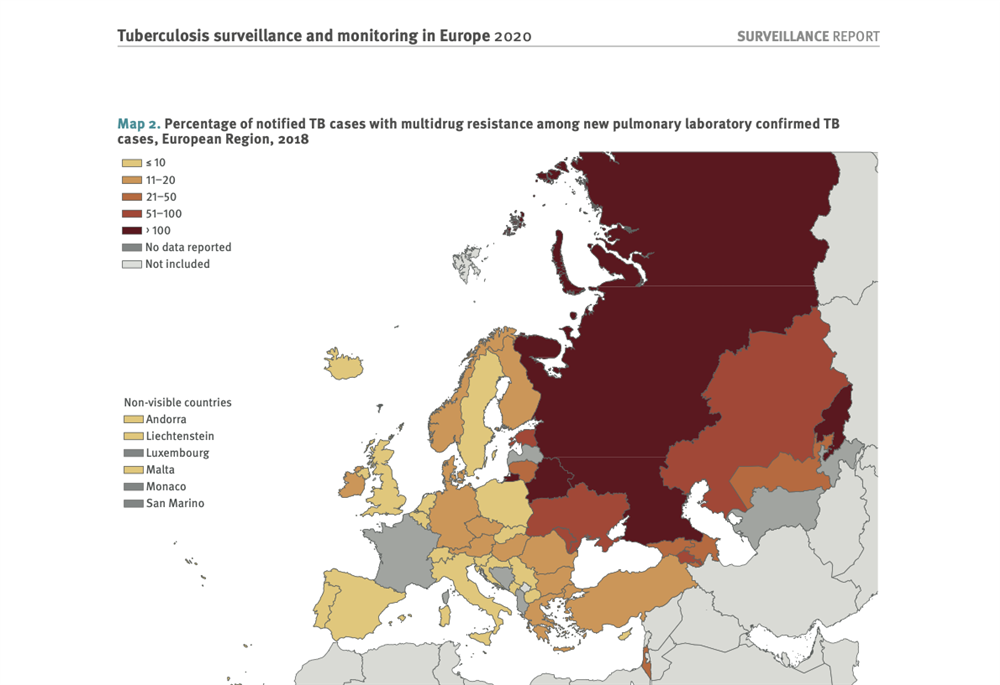
MDR-TB prevalence among new cases in most EECA countries where TB-REP 2.0 operates is critically high: Georgia – 11.5%, Kazakhstan – 16%, Moldova – 24.4%, Ukraine – 22.3%, Tajikistan – 72.7%, Belarus – 36.6% and Kyrgyzstan – 30.4%.
 “This is a breakthrough that will boost the transition to all-oral shorter treatment regimens for DR-TB in line with latest WHO policies and support high MDR-TB burden countries achieve better treatment outcomes. Low- and middle-income countries in our region now have the opportunity to substantially scale up and provide treatment to 32% more patients with the existing resources, » says Liliana Caraulan, TB-REP 2.0 Program Coordinator (PAS Center). – “A great collaboration between Stop TB Partnership, Johnson&Johnson, USAID and Global Fund that will save lives”.
“This is a breakthrough that will boost the transition to all-oral shorter treatment regimens for DR-TB in line with latest WHO policies and support high MDR-TB burden countries achieve better treatment outcomes. Low- and middle-income countries in our region now have the opportunity to substantially scale up and provide treatment to 32% more patients with the existing resources, » says Liliana Caraulan, TB-REP 2.0 Program Coordinator (PAS Center). – “A great collaboration between Stop TB Partnership, Johnson&Johnson, USAID and Global Fund that will save lives”.
Note that the World Health Organization (WHO) recommends the oral drug bedaquiline as the basis for the treatment of MDR-TB in its updated manual, published June 15, 2020, to replace older and more toxic drugs that must be injected daily. After all, injection treatment requires people to go to medical facilities. The older, longer MDR-TB treatment used in many countries to date has required people to take up to 14,000 tablets for almost two years and endure up to eight months of painful daily injections!
The latest WHO 2020 Compendium of Guidelines for the treatment of drug-resistant tuberculosis recommends a new shorter, completely oral regimen for use in operational research for patients with MDR-TB and additional resistance to fluoroquinolones. This regimen lasts 6–9 months and contains a new compound that has not previously been used in treatment regimens for tuberculosis — prometanide in combination with bedaquiline and linezolid.
According to WHO, this will lead to a significant improvement in treatment outcomes and the quality of life of people with TB. Also, in light of the COVID-19 pandemic, WHO experts further recommended that countries treat people with MDR-TB not in a hospital setting, but at home, with all the necessary precautions.
Thus, a challenge of particular importance to governments in Eastern Europe and Central Asia, at a reduced price and in the context of COVID-19 challenges for people with TB, is to increase the use of bedaquiline as the main part of all oral treatment regimens for DR-TB. This is a new major challenge in advocating for increased access to treatment for NGO activists and patient organizations in the EECA region.
It is worth noting that the price of J&J for bedaquiline remains a critical barrier for countries expanding this critically important and effective treatment. Given the substantial taxpayer funding that J&J received for research and development of this drug from the United States (USA) and other countries, activists from around the world called on the corporation to lower the price to increase access to treatment. A study by scientists from the University of Liverpool showed that bedaquiline can be produced and sold for only $0.25 a day.
The new reduced price is 32% lower than the previous lowest price available for the list of countries identified by J&J as low- and middle-income countries, which include countries in the EECA region linked to procurement commitments made through the Global Medicines Fund (GDF), an organization managed by the Stop TB Partnership, which coordinates the global fight against tuberculosis.
At the same time, the whole world is eagerly awaiting the appearance of more accessible generic versions of bedaquiline.
* Calculations are based on a 32% reduction over the previous price of $400 for a six-month course of treatment